جستجو
سبد خرید (0)
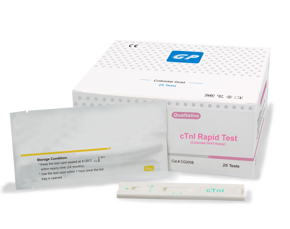
تست تشخیص سریع کیفی تروپونین I
تست سریع تشخیص تروپونین I
تست سریع تشخیص تروپونین I
Colloidal Gold Assay
نمونه : خون کامل، سرم، پلاسما cut off : 0.5 ng/mL
تست تروپونین Getein Biotech Inc. یک تست سریع ایمونوکروماتوگرافیک جهت تشخیص تروپونین I قلبی در نمونه های پلاسما، سرم و خون کامل بیماران می باشد. این کیت جهت استفاده به عنوان روش کمکی در تشخیص سکته های قلبی ارائه شده است. این آزمایش در تشخیص به روش (in vitro) بیماری مذکور به کار برده می شود. نتایج مثبت نشان دهنده خطر بالای سکته قلبی می باشد. با وجود این، نتایج منفی دلیل بر قطعیت عدم وجود سکته قلبی و عدم انجام سایر آزمایشات مربوط به پیگیری سکته قلبی نمی باشد.
دانلود کاتالوگ$0.00